Is Victory in Sight?
Covid-19 vaccine rolled out
An outcome of joint research efforts by an American pharmaceutical giant and a German biotechnology company, Pfizer-BioNTech Covid-19 vaccine got an emergency use authorization (EUA) from the Food and Drug Administration (FDA) of the US on 11 December for the prevention of Covid-19 in individuals 16 years of age and older. This EUA allowed the Pfizer-BioNTech Covid-19 vaccine to be distributed in the United States. The vaccine authorization is being considered by many as a step which can “alter the course of the pandemic” in spite of the fact that EUA is not an approval, and is issued by the FDA in an emergency when there are no adequate approved or available options for treatment of life-threatening diseases. This implies that the needed evidence for a full FDA approval of the Pfizer-BioNTech Covid-19 vaccine is still not there.
Is this really the beginning of the last chapter of the book?
I argue that calling this vaccine launch as the “last chapter” is tantamount to declaring victory too soon. There are several drawbacks of EUA which need to be understood before considering this rollout of the vaccine as a decisive blow to Covid-19.
By putting the Pfizer-BioNTech Covid-19 vaccine into the American health system, the FDA has allowed the availability of a treatment that is yet to be fully tested, and possess the required evidence for its full approval. How much more time is needed for collecting that evidence and data before a full approval is granted is not clear to anyone. Moreover, if at any time during this ensuing period, this vaccine creates any health concerns, the subsequent public trust in the entire EUA process will erode substantially.
The FDA has committed a blunder before. In October, through the EUA process, it approved remdesivir, an antiviral, for treatment of the people hospitalized with Covid-19. This was done hurriedly before any clinical data was published. World Health Organization (WHO), on the contrary, through a study on more than 11,000 participants, found remdesivir to have little effect on how long people stayed in hospital and no effect on death.
A similar scenario was witnessed in the case of hydroxychloroquine, a drug used to treat malaria. A small study had touted hydroxychloroquine as an effective treatment of Covid-19 and it was approved through EUA. Later investigations concluded that it lacked the required efficacy against Covid-19 and the risks of using it for Covid-19 outweighed its benefits, if any. This led to the revocation of the EUA earlier granted to hydroxychloroquine by the FDA.
These examples of hasty approvals, and subsequent revocation in the case of hydroxychloroquine, surely led to confusion and mistrust in specifically the treatments and the regulatory process of the FDA in general. One bad story like this on Pfizer-BioNTech Covid-19 vaccine, everything will fall apart!
A nationally representative survey conducted by McKinsey and Company clearly shows how America perceives this new vaccine and the EUA. More than half the respondents of this survey reported that they are likely to delay or decline vaccination despite the regulatory authorization. Their main concern for the vaccine hesitancy was its safety. As many as 26 percent believed that the development of the vaccine was too rushed with remarks like, “Nothing is guaranteed in a rushed vaccine” and “I am worried about the effectiveness.” Many side effects lasting for several days like pain at the injection site, tiredness, headache, muscle pain, chills, joint pain and fever have already been reported, especially after the administration of the second dose.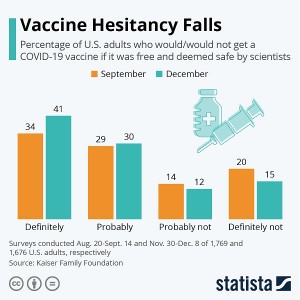
It is not only the public trust at stake here. The Journal of American Medication Association (JAMA) identifies this fast-tracking of treatments with the risk of non-optimal therapies being administered as well as sucking up hospital resources and aggravating health inequities.
There is the example of a monoclonal antibody therapy by pharmaceutical company Regeneron. It could help boost the immune system in fighting Covid-19 but its distribution and administration could tax outpatient clinics. Currently studied monoclonal antibody preparations require a one-hour intravenous infusion and the medical infrastructure that can provide infusions of therapeutics generally do so in dedicated facilities previously reserved for delivery of immune-suppressing biologic agents and chemotherapy.
Another apprehension is regarding the nearly 8 million US residents who have lost their employer-sponsored health insurance due to the pandemic, worsening disparities in insurance coverage for Black and Latin population, and leaving those with the highest rates of infection unable to afford the best possible treatment.
Coming days are not going to be easy for either America or the world at large. Social distancing, and not vaccines, as practiced by many countries that have controlled infection rates will remain the most powerful tool to curb the spread of Covid-19 for at least a couple of more years. There are presently 13 manufacturers in different parts of the globe who have entered into premarket purchase commitments for at least 7.48 billion doses, or 3.76 billion courses. A very recent research published in BMJ (formerly the British Medical Journal) by a Professor at the Johns Hopkins University shows that even if all 13 of these vaccine manufacturers were to succeed in reaching their maximum production capacity, at least a fifth of the world’s population would not have access to vaccines until 2022. Victory against the deadly Covid-19 can only be achieved through a multi-pronged strategy where the highest priority must be given to social distancing.
The author is a civil servant presently pursuing a doctorate in technology and innovation policy
Email: atharmansoor74@gmail.com
 Jahangir's World Times First Comprehensive Magazine for students/teachers of competitive exams and general readers as well.
Jahangir's World Times First Comprehensive Magazine for students/teachers of competitive exams and general readers as well.